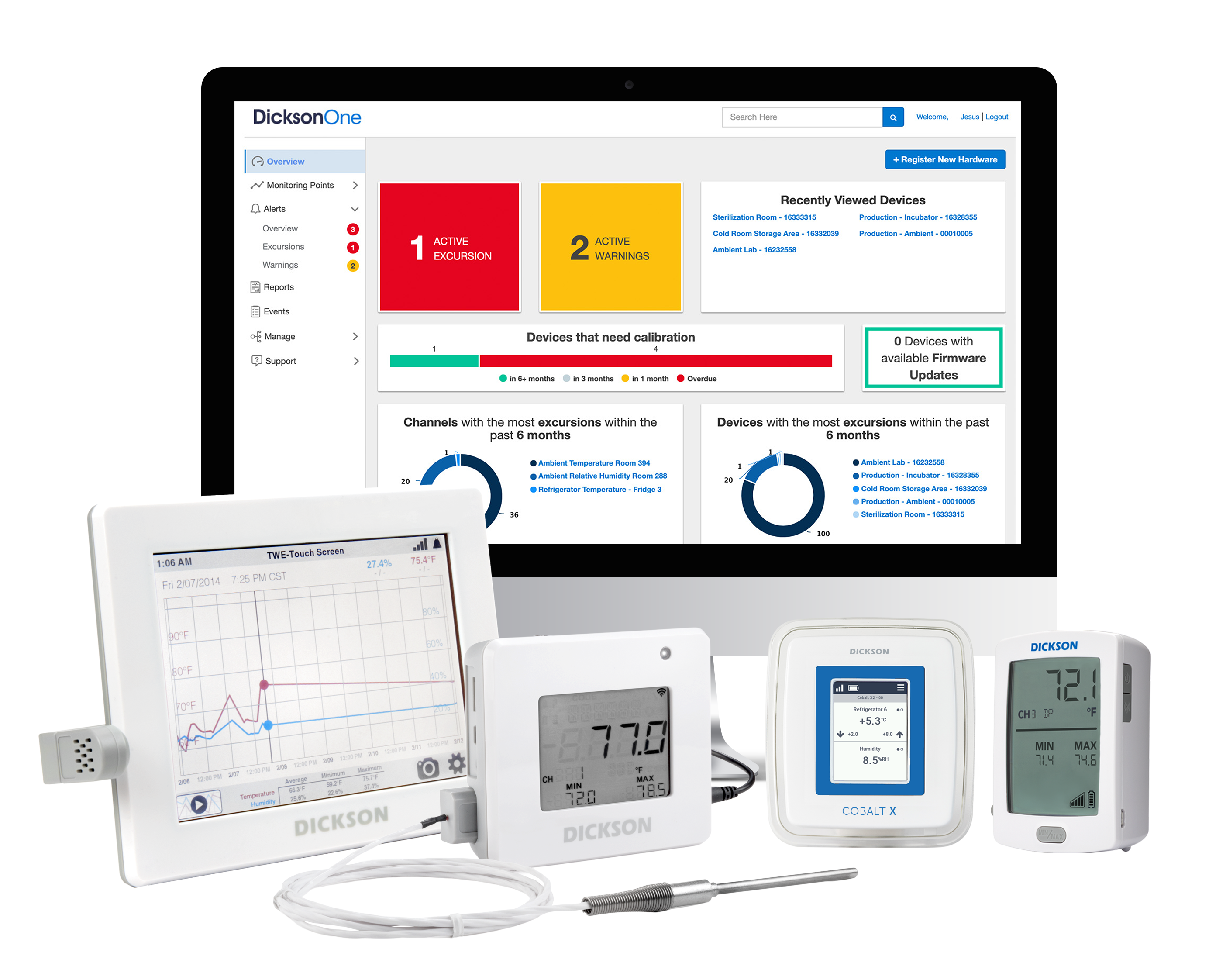
In 2019, Oceasoft and Dickson merged and began to combine their expertise and resources. The two companies have formed a powerful alliance offering unparalleled capabilities in data collection, analysis, and reporting. Dickson Data's extensive experience in temperature and humidity monitoring and...
Read Moreenvironmental monitoring
Poor storage conditions that expose delicate pharmaceuticals and biologics to changes in temperature and humidity can be very costly in the form of lost product, poor end quality for patients, and damage to reputation.
What Defines Pharmaceutical Product Quality?
Pharmaceutical products must be...
Read MoreA growing trend in the pharmaceutical industry is the development of more complex and fragile products, like biologicals, chemical mixtures, and large molecules (COVID-19 vaccines are an example of this trend).
Read MoreSince their introduction, IoT devices have evolved from technical curiosities to ubiquitous elements in daily life, finding applications in home automation, healthcare, manufacturing, agriculture, traffic control, and a diverse collection of other areas.
Read MoreA central aspect of doing business in highly regulated industries, like pharmaceutical manufacturing and distribution, healthcare, or aerospace, is the need to have extensive systems in place to protect consumers and maintain regulatory compliance. That includes systems to monitor the environmental...
Read MoreCold chains are specially designed distribution networks in which product temperature is controlled and monitored from end to end. Many types of products are distributed using cold chains, including produce, frozen foods, specialty chemicals, and the focus of this article, pharmaceuticals.
Read MoreAdvances in communications technology over the last decade, especially in wireless communication, have enabled the widespread use of cloud-based data storage in the pharmaceutical/biotech and healthcare industries.
Read MoreMany over-the-counter and prescription drugs can be stored under normal, ambient conditions. As long as the temperature is reasonably controlled, humidity is low, and they’re kept out of direct sunlight, they have a long shelf life.
Read MoreEnvironmental monitoring is important in several aspects of healthcare facility operations. For example, the temperature and humidity in critical care spaces, like operating rooms and ICUs, must be controlled for patient safety and comfort, and to maximize energy efficiency. Continuous monitoring...
Read MoreAn early 2021 inspection of an Eli Lilly pharmaceutical plant in Indianapolis led to the FDA issuing a Form 483. The inspectional observations that were listed in the form included problems with environmental monitoring, microbial contamination prevention procedures, and root cause investigations...
Read More